 
Most chaperones interact with multiple co-chaperones, and co-chaperones can interact with multiple chaperones, thereby modifying their function or the fate of their substrates. The chaperone system is highly versatile. Folding enzymes that catalyze folding-accelerating reactions, such as peptidyl prolyl cis– trans isomerization or protein disulfide isomerization 16, 17, are also considered as chaperones. Families of co-chaperones modulate the activity of main chaperones by regulating their ATPase cycle or the recognition, binding, or release of chaperone substrates, and include HSP10 9, HSP40 (DNAJ) 13, nuclear exchange factors (NEFs) 14, and co-HSP90 15. Families composing the main chaperone machinery, which modulate protein structure without participating in the final protein complex, include prefoldin 7, the small heat shock proteins (sHSP) 8, and the main ATP-hydrolyzing chaperones, HSP60 9, HSP70 10, HSP90 11, and HSP100 12. 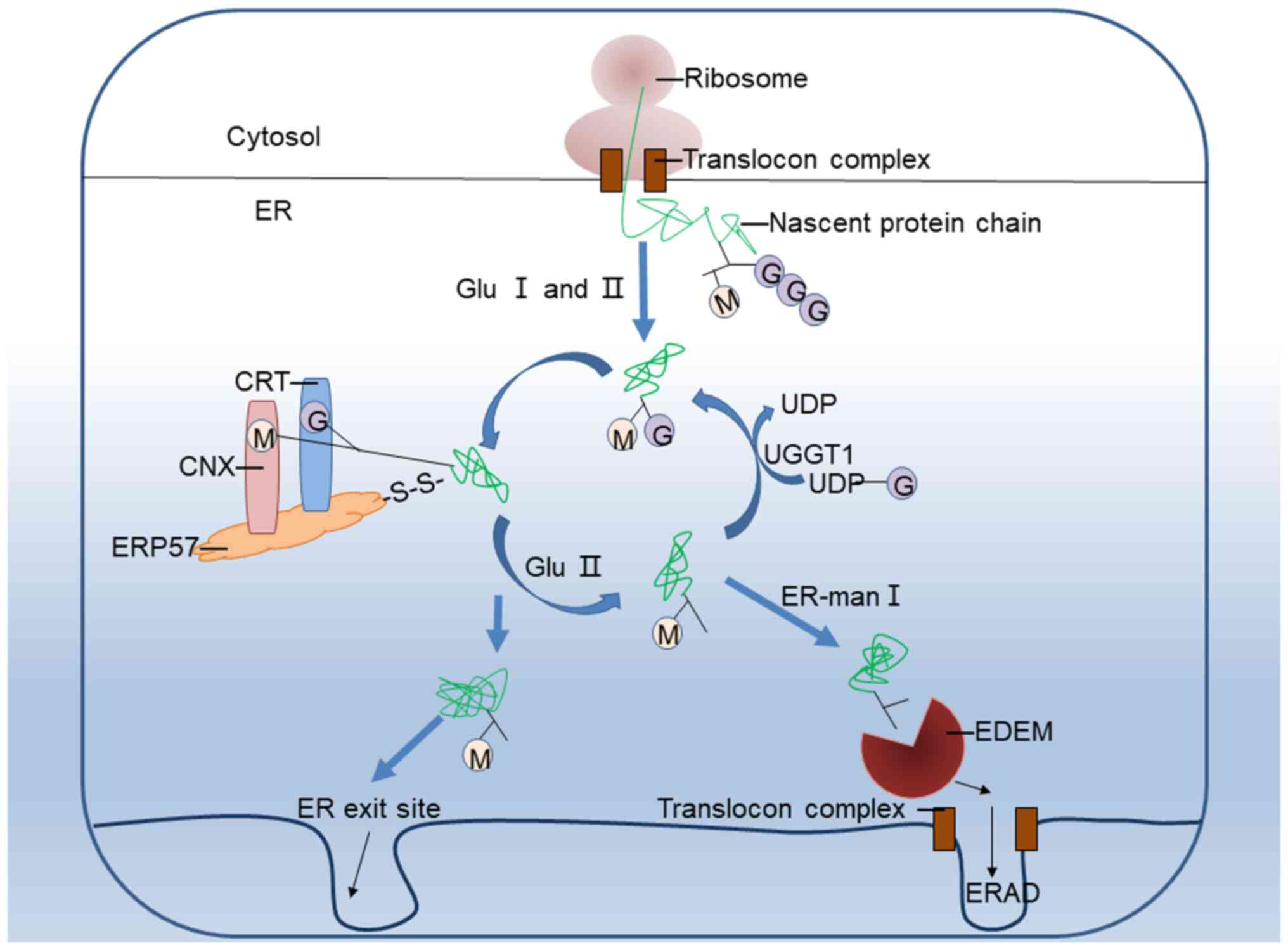
In human cell lines, for example, they were shown to compose ~10% of the total proteome mass 6.Ĭhaperones have been grouped into families based on their molecular mass, common domains, protein structure similarity, and common function 1. In accordance with their fundamental roles, chaperones are abundant proteins. Across species, they promote de novo protein folding and protein maturation 1, protein translocation 2, protein-complexes assembly and disassembly 3, protein disaggregation and refolding 4, and protein degradation 5. In this work, we expand the known functional organization of de novo versus stress-inducible eukaryotic chaperones into a layered core-variable architecture in multi-cellular organisms.Ĭhaperones are highly conserved molecular machines that control cellular protein homeostasis (proteostasis). Analysis of human organ development and aging brain transcriptomes reveals that these functional networks are established in development and decline with age. Together with variable chaperones, they form tissue-specific functional networks. Core chaperones are significantly more abundant across tissues and more important for cell survival than variable chaperones. We demonstrate via a proteomic analysis that the muscle-specific signature is functional and conserved. 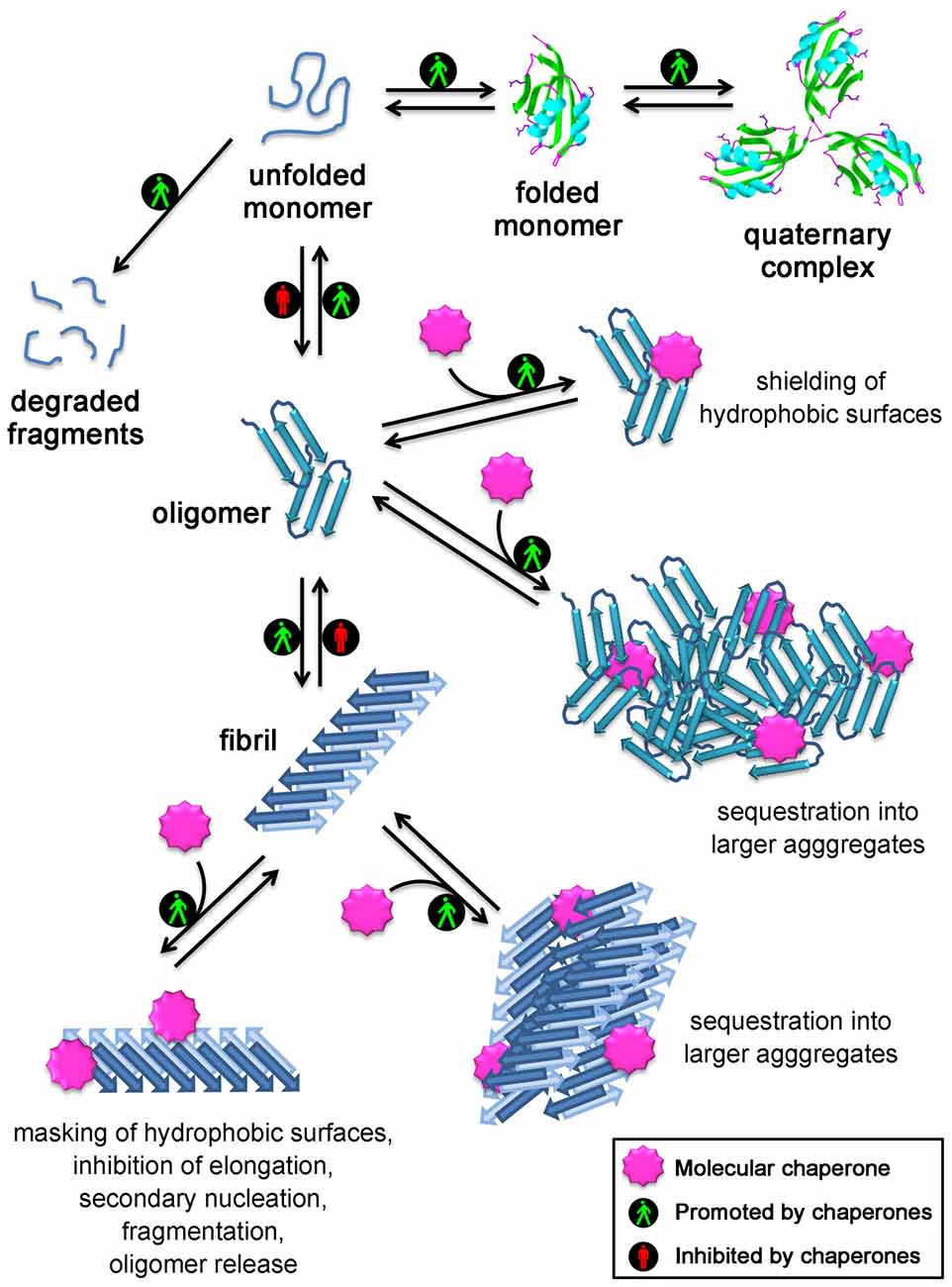
Through computational analyses of large-scale tissue transcriptomes, we unveil that the chaperone system is composed of core elements that are uniformly expressed across tissues, and variable elements that are differentially expressed to fit with tissue-specific requirements. Yet, the organization of the chaperone system across physiological human tissues has received little attention. The sensitivity of the protein-folding environment to chaperone disruption can be highly tissue-specific. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
